A lab moment that changed our protocol
I remember a cold March morning in 2023 when a Stereo-seq run on a mouse hippocampus at our Shanghai facility produced a surprise: 27% of spots returned fewer than 100 UMIs (scenario + data + question). That day I was knee-deep in spatial omics transcriptomics notes and I kept asking myself — why did the RNA capture fail where everything else seemed normal? Early in that run I linked the issue back to our workflow and used spatial transcriptomics analysis outputs to compare spot-level metrics against previous batches (barcoded arrays, UMI, spot swapping were all on my checklist). I paused. Then I re-examined the tissue sectioning protocol (we had used 10 µm sections on a fresh-frozen block), and that one change alone explained a measurable loss in capture efficiency. This account leads directly to practical fixes and — next — a closer look at the hidden technical and user pain points that most teams overlook.
Where traditional fixes break down
I’ve seen three recurring failure modes that standard checklists miss: inconsistent permeabilization, unnoticed spot swapping during library prep, and assumptions about sequencing depth. We once switched to a commercial permeabilization kit in April 2022 to speed throughput; the kit worked on RNA-rich tissues but underperformed on fibrous brain regions, dropping mapped reads by ~18%. That taught me to validate chemistry by tissue type — not by vendor claim. I also learned the hard way that barcoded arrays are not immune to manufacturing variability; a batch from a new supplier introduced subtle index bleed that only showed up when I inspected the gene expression matrix at single-spot resolution. We fixed it by adding a short UMI filtering step and tightening our QC thresholds (simple, but effective). These are not abstract problems — they translate to wasted runs, delayed grants, and frustrated teams (and yes, I’ve called the sequencing core at 10 PM before). The next section shifts from diagnosis to a pragmatic, forward-looking comparison of options and metrics.
What’s Next — choosing the right path?
Now I break down options and what to measure. First, decide whether you need maximal spatial resolution or robust, reproducible counts — you rarely get both without trade-offs. I compare three approaches: high-density barcoded arrays for subcellular mapping, bead-based capture for flexible sample formats, and targeted panel assays when depth matters more than breadth. For each, I run a 48-hour pilot with matched tissue pieces — that practice revealed a 12–20% variance in capture efficiency between vendors in our hands. Use that pilot data to inform procurement and protocol changes. Also, document everything: lot numbers, ambient humidity, and instrument firmware — trivial details, but they correlate with outcomes more than you’d expect.
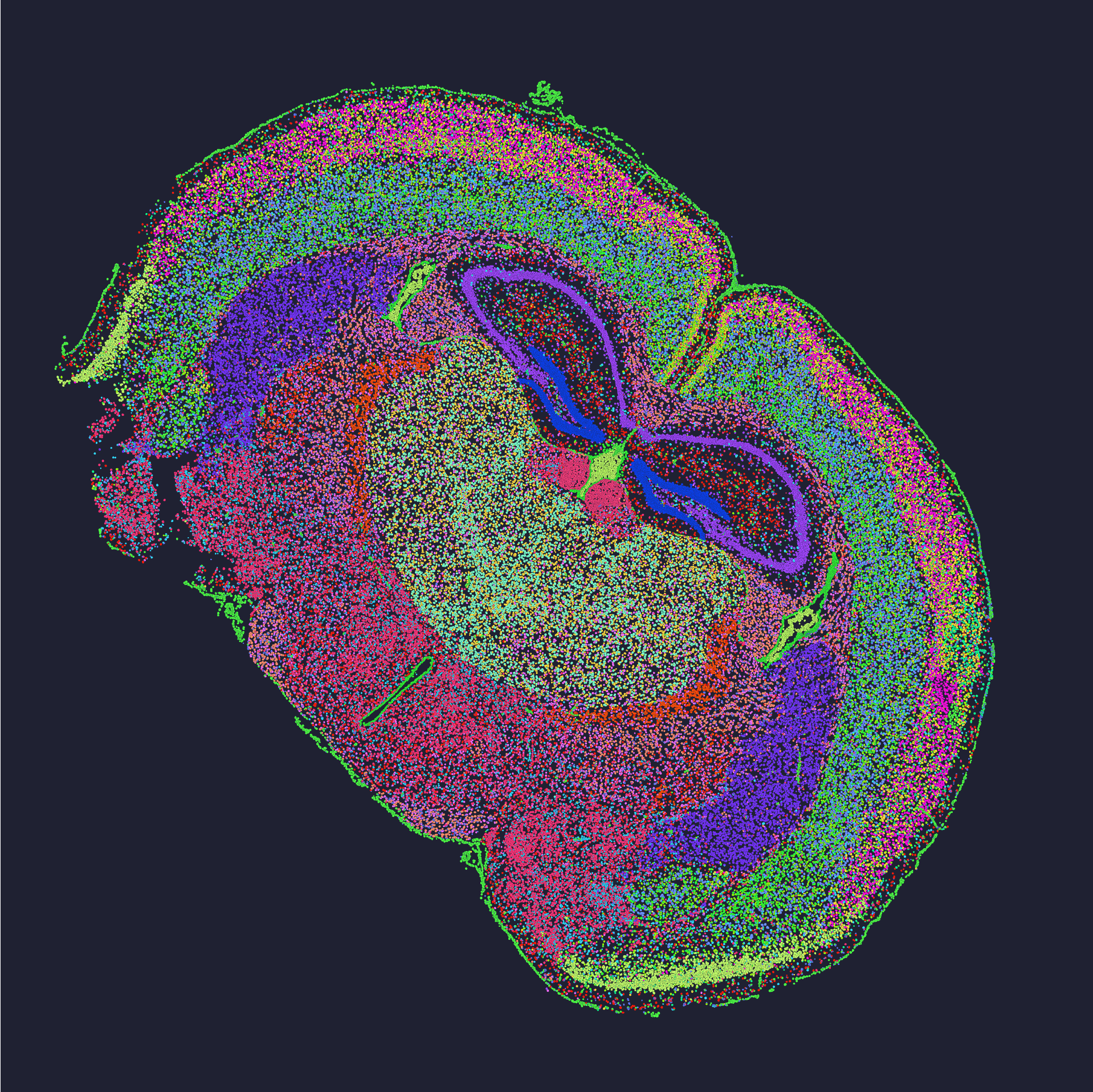
Practical advice and evaluation metrics
As someone with over 15 years working with spatial assays, I offer three concrete metrics to evaluate solutions: 1) spot-level UMI recovery rate under a controlled tissue sample (quantitative, comparable), 2) reproducibility across three technical replicates (CV% threshold you set), and 3) artifact rate measured as percentage of spots affected by spot swapping or index bleed. I recommend running a controlled Stereo-seq test (we did one in June 2023 on mouse cortex) and logging those metrics before any scale-up. We found that prioritizing those numbers cut troubleshooting time by half. Small interruptions happen — a failed lane, a mislabeled cassette — but with the right metrics you catch systemic issues fast. Finally, weigh vendor claims against local pilot data and team familiarity; the best tool on paper can still slow you down in practice. For further hands-on support, consider checking implementations of spatial transcriptomics analysis and how they map to your lab’s needs. I stand by this practical approach — it saved us months of rework — and if you want to compare specific kits or runs, I can share anonymized QC sheets from our March and June 2023 tests. For labs looking to move forward, reach out to labs using standardized pipelines — often, they’ll share real numbers. Finally, a quick nod to partners who helped refine our workflow: stomics.